
While holding hot objects, it feels hot. This phenomenon occurs due to heat transfer from the hot body to our skin because of Conductive heat Transfer. This rate of conductive heat transfer depends on material thermal properties, temperature difference, and area of contact.
In this article, we will discuss What is conduction Heat Transfer? and how to calculate Conduction heat Transfer? We suggest you also read this article on various modes of heat transfer.
What is Conductive Heat Transfer?
Conduction Heat Transfer is the transfer of heat within thermally conductive or thermally contacted bodies due to temperature differences.
It occurs in solid bodies due to molecular excitement or lattice vibrations of free electrons. Whereas in liquids, conductive heat transfer occurs due to molecular movements, and in gases, it occurs due to molecular collision.
Examples of Conduction Heat Transfer
Following are the examples of conduction heat transfer in products that we use in our day-to-day life.
- Transfer of heat from the burner to the metal pan.
- Hands get burned if we touch hot surfaces due to conductive heat transfer from hot surfaces to our skin.
- Ironing of clothes.
- Transfer of heat from the heat-dissipating electronics components to the heatsink. Click this link to know more about the transfer of heat in a heat sink.
- The car engine gets hot when turned on.
- The cup becomes hot when hot coffee is poured into a cup.
Fourier’s Law (Conduction Heat Transfer Calculation)
The rate of conduction heat transfer is directly proportional to the contact area, material thermal conductivity, temperature difference, and inversely proportional to the thickness.
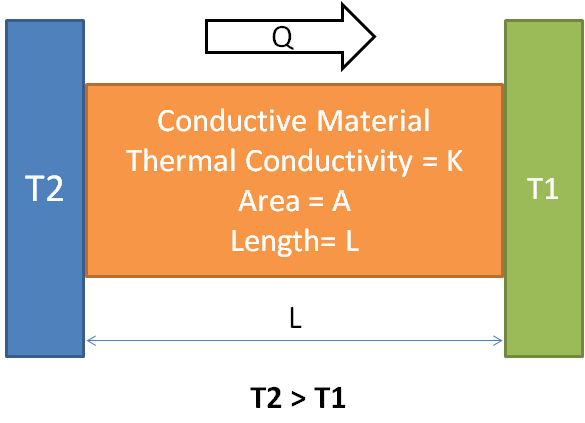
Conduction heat Transfer Equation
Conductive Heat transfer (Qc) = – K A (dT / L)
Where
Qc = Conductive heat transfer per unit time in Watt
A = cross-section area in square meter
k = Material thermal conductivity (W/mK)
dT = Temperature differential
L = Thickness or length of the part in meter
Where (dT / L) is known as temperature gradient. Its value is always negative because the temperature gradient vector is in the opposite direction of heat flow.
Conduction heat transfer is more significant in solid bodies and it follows the first law of thermodynamics.
Thermal Conductivity of a Material
| Thermal Conductivity (These values are for Indicative Purpose Only) | |
|---|---|
| Material | Thermal Conductivity (W/mK) |
| Copper | 384 |
| Aluminum | 205 |
| Iron | 79 |
| Water | 0.6 |
| Air | 0.026 |
| Silicon | 0.01 |
| Diamond | 895 |
| Bakelite | 0.14 |
Thermal Insulation
Thermal insulation is a property of a material that is inversely proportional to thermal conductivity. In other words, the higher the thermal insulation, the lower will be the thermal conductivity. Therefore insulation materials are used to reduce conductive heat transfer.

Low thermal conductivity or high insulation materials are used to reduce heat transfer from one material to another.
For example, a Heating Pan Handle is manufactured in Bakelite material because bakelite has high thermal insulation properties.
Relationship in thermal and electrical Conductivity of a Material
There is no direct relationship between the electrical and thermal conductivity of a material. A good conductor of electricity doesn’t need to be a good conductor of heat as well. For example, a diamond is a good conductor of heat but a bad conductor of electricity.
Conductive Heat Transfer Calculator
Conductive Heat Transfer Problem Examples
Question: Calculate the difference between the rate of heat transfer from 1000 mm² area and 2mm thickness Bakelite and Aluminum wall considering 80°C temperature difference.
Answer:
Temperature difference between two walls (dT) = 80°C
Wall thickness (L) = 2 mm = 0.002 mm
Cross Section Area (A) = 1000 mm² = 0.001 Meter²
Bakelite thermal conductivity (k1) = 0.14 W/mK
Aluminum thermal conductivity (K2) = 205 W/mK
Rate of Heat Transfer (Bakelite) = -KA (dT/L) = 0.14 × 0.001 × (80/0.002) = 5.6 Watt
Rate of Heat Transfer (Aluminum) = 205 × 0.001 × (80/0.002) = 8200 Watt
Therefore the rate of heat transfer in aluminum is higher than in Bakelite. You can also use the above calculator for this calculation.
We will keep adding more information on conductive heat transfer and how to calculate it. Add your suggestions, comments, or questions on conduction heat transfer in the comment box. We suggest you also read these articles on convection and radiation heat transfer.
Add a Comment